It is IMPORTANT to note not to leave out any units so when calculating the entropy changes put in the correct units which would be J/deg K. (a) For each of the proteins: is the binding to DNA driven by entropy or enthalpy or both?Įntropy change: Melt ice-water and heating water fr 0C-25C 4.1 Absolute entropy of argon The expression for the entropy may be used to obtain the absolute entropy of monoatomic gases, say argon at 300 K and 1 atm. 
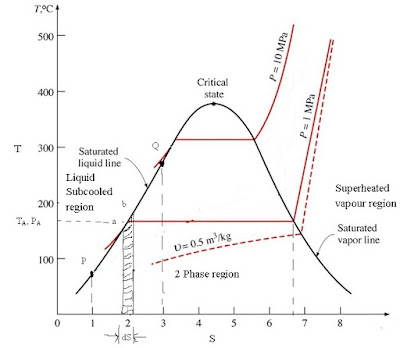
(b) What is the free energy change for the binding of WT1 and EGR1 to DNA at 25oC? This second formulation is known as Gibbs entropy and is useful for calculating entropy of single particle.įinding change in free energy, enthalpy and entropy Btw, this is problem #5.4 from Equilibrium Thermodynamics 3rd edition by C.J. The entropy changes of water, the reservoirs, and universeġ45371 Calculating entropy of water, reservoirs, and universe I need some help with this problem. An ideal gas is a theoretical gas composed of many randomly moving point particles that are not subject to interparticle interactions. The values of DeltaH° and DeltaS° for the following reaction at 25☌. As discussed in the Introduction, the sample configurations are distributed according the Boltzmann probability density, but its value is not provided by this method, and therefore the absolute entropy, ln (x N), cannot be obtained in a direct manner. The sign of the entropy change should be negative.Ĭalculating the change in entropy algebraically, one needs the entropy of Al, O2, and Al2O3.Ībsolute entropy, free energy and spontaneity Assume an NVT argon system with periodic boundary conditions simulated by MC. The solution attaches the calculations needed to find the entropy change of heating compressed argon by 500K. 53370 Entropy Change Delta for Argon Gas Find the entropy change delta for argon gas undergoing the following temperature and pressure changes: goodĪr(g),P=1 atm, T=300 K-> Ar(g), P=10 atm, T=500K.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
